Dr. Peter Attia is quietly returning to public life, publishing substantive medical analysis for the first time in weeks after a stretch that included a very public fallout from his newly announced CBS News contributor role and the release of documents linking him to Jeffrey Epstein.
The longevity physician and bestselling author had barely settled into his role at the network when the ground shifted beneath him. On January 27, 2026, CBS News announced Attia as one of 19 new contributors. Within days, the U.S. Department of Justice released another tranche of documents from its Epstein investigation, and Attia’s name appeared in those files hundreds of times.
He did not wait long to respond. In a public statement, Attia described the emails as “embarrassing, tasteless, and indefensible,” while making clear he had no involvement in any criminal activity. He stated he never flew on Epstein’s plane, never visited his island, and never attended “any s*x parties.”
His exit from CBS followed shortly after. A spokesperson for Attia explained that the role “was newly established and had not yet meaningfully begun,” and that Attia “stepped back to ensure his involvement didn’t become a distraction from the important work being done at CBS.” The network declined to comment publicly beyond that.
For someone whose brand is built on trust and the long view on health, the reputational damage was measurable, if not catastrophic. On YouTube, Attia lost roughly 30,000 subscribers, and his view counts have trended downward, though it remains too early to assess the full trajectory.
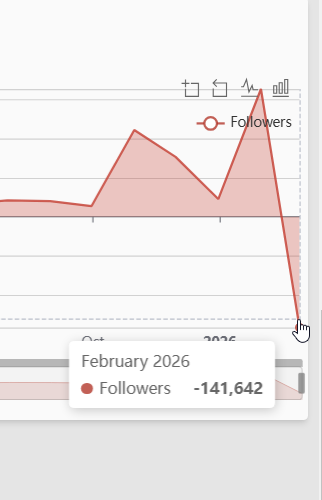
On Instagram, the losses were more pronounced: approximately 150,000 followers, representing close to 10 percent of his audience. He still holds 1.6 million followers on the platform.
Now, quietly and without fanfare, Attia appears to be resuming work. His latest newsletter, co-authored with Taylor Yeater and Nicholas Nelson, arrived on March 7, 2026, and it is very much in the spirit of what built his following in the first place: dense, methodologically careful analysis of a contested medical question.
The subject is GRAIL’s Galleri test, a blood-based multi-cancer early detection tool that has been commercially available in the United States since 2021 at a list price of $949. The test screens for signals associated with more than 50 cancer types using cell-free DNA analysis. When the NHS released trial results suggesting Galleri had missed its primary endpoint, headlines declared the technology essentially finished and GRAIL’s stock fell roughly 50 percent overnight.
Attia’s read is more restrained. He notes the data came through a corporate press release, not a peer-reviewed publication, meaning the full dataset has not yet been publicly presented. Full results are expected at the American Society of Clinical Oncology annual meeting in late May or early June 2026.
“All of that is pending and, until it is available, we must wait before making strong conclusions in either direction,” he writes.
He also points to a potentially important signal buried within the trial’s results. Among 12 specific cancer types, including pancreatic, ovarian, and liver cancers, Stage IV diagnoses appeared to decrease with each round of annual screening, with reductions exceeding 20 percent in rounds two and three. Stage I and Stage II diagnoses increased in the intervention group, and the overall cancer detection rate was reportedly four times higher than standard care alone.
His argument is not that the technology has proven itself, but that the coverage has outpaced the evidence. He writes that the composite primary endpoint, which combined Stage III and Stage IV reductions, may have obscured a clinically meaningful improvement in Stage IV disease specifically. Had Stage IV reduction alone been the pre-specified measure, the story might have looked different. He is careful to note that re-framing endpoints after the fact is not how clinical trials work, but the distinction matters for understanding what the data actually show.

